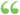 As used in this subsection and subsection (e), the term "substantial evidence" means evidence consisting of adequate and well-controlled investigations, including clinical investigations, by experts qualified by scientific training and experience to evaluate... As used in this subsection and subsection (e), the term "substantial evidence" means evidence consisting of adequate and well-controlled investigations, including clinical investigations, by experts qualified by scientific training and experience to evaluate...  Committee Prints - 411. lappuseautors: United States. Congress. Senate. Committee on Labor and Public Welfare - 1969Pilnskats Committee Prints - 411. lappuseautors: United States. Congress. Senate. Committee on Labor and Public Welfare - 1969Pilnskats - Par šo grāmatu
 | United States - 1977 - 1276 lapas
...adequate and well-controlled Investigations, including clinical investigations, by experts qualified by tively, of the agreement, is authorized and directed...the United States from time to time, at par, and responsibly be concluded by such experts that the drug will have the effect it purports or is represented... | |
 | United States - 1964 - 1098 lapas
...adequate and wellcontrolled investigations, including clinical investigations, by experts qualified by scientific training and experience to evaluate the...drug involved, on the basis of which it could fairly TITLE 21.— POOD AND DRUGS Page 1142 and responsibly be concluded by such experts that the drug will... | |
 | 1980 - 684 lapas
...and well-controlled investigations, including clinical (field) investigation, by experts qualified by scientific training and experience to evaluate the...the conditions of use prescribed, recommended, or suggested in the proposed labeling. (ii) The following principles have been developed over a period... | |
 | 1996 - 618 lapas
...and well-controlled investigations, including clinical (field) investigation, by experts qualified by scientific training and experience to evaluate the...the conditions of use prescribed, recommended, or suggested in the proposed labeling. (ii) The following principles have been developed over a period... | |
 | 2000 - 698 lapas
...training and experience to evaluate the effectiveness of the new animal drug involved that the new animal drug will have the effect it purports or Is represented...suggested in the labeling or proposed labeling thereof. Substantial evidence shall include such adequate and well-controlled studies that are, as a matter... | |
 | 2002 - 628 lapas
...training and experience to evaluate the effectiveness of the new animal drug involved that the new animal drug will have the effect it purPorts or is represented...suggested in the labeling or proposed labeling thereof. Substantial evidence shall include such adeluate and well-controlled studies that are, as a matter... | |
 | 1969 - 668 lapas
...evidence available when the application was approved, that there is a lack of substantial evidence that the drug will have the effect it purports or...prescribed, recommended, or suggested in the labeling thereof; or (4) That the application contains any untrue statement of a material fact; or (c) The Commissioner... | |
 | 1980 - 718 lapas
...available to him when the application was approved, there is a lack of substantial evidence that such drug will have the effect it purports or is represented...prescribed, recommended, or suggested in the labeling thereof. (4) That any nonclinical laboratory study contained in the application was not conducted in... | |
 | 1988 - 772 lapas
...available to him when the application was approved, there is a lack of substantial evidence that such drug will have the effect it purports or is represented...prescribed, recommended, or suggested in the labeling thereof. (4) That any nonclinical laboratory study that is described in the application and that is... | |
 | 1977 - 568 lapas
...(5) There is a lack of substantial evidence based upon adequate and well-controlled investigations that the drug will have the effect it purports or...the conditions of use prescribed, recommended, or suggested in the proposed labeling thereof. An adequate and well-controlled investigation must satisfy... | |
| |